Confidence in the Classroom
Make Quadrant’s COVID-19 PCR Saliva Test part of your health & safety plan.
A simple saliva test for COVID
Unlike nasal swabs, this is a simple, non-invasive, easy-to-use, saliva collection device. Students from kindergarten through their senior year can easily self-collect their samples.
Our saliva test accurately detects all known variants of COVID-19.

An accurate test matters.
The FDA requires that manufacturers measure the sensitivity of their test against a standardized SARS-CoV-2 Reference Panel. The measure of this test sensitivity is known as the Limit of Detection (LOD). High LODs will miss more infected patients and result in more false negatives.
Quadrant’s COVID-19 PCR Saliva Test has the lowest LOD of any saliva test currently on the market and remains effective for detecting COVID variants.
Our test has been used to at more than 100 universities, 400 primary school districts, and municipalities across the Northeast.
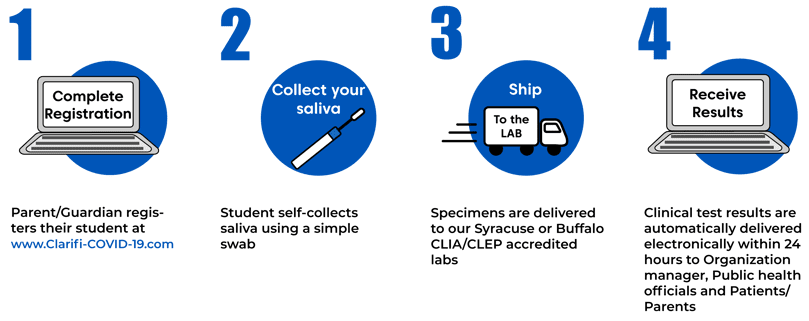
The collection process is quick and easy.
Stay Vigilant
Regular COVID testing helps you confidently keep your schools open and provides your students, staff, and faculty with a safe learning and working environment.
Ordering Quadrant’s COVID-19 PCR Saliva Test is simple. Request more information below and a member of our team will reach out shortly.
Getting Started
In order to initiate COVID-19 PCR Saliva testing with Quadrant Laboratories, school officials must complete the following forms:
Additional guidance on testing procedures and guidelines can be found here:
*On September 22, 2020, the Clarifi® COVID-19 Test Kit obtained Emergency User Authorization (EUA) by the Food and Drug Administration (FDA) to be used for the diagnosis of SARS-COV-2. The Clarifi COVID-19 Test Letter of Authorization, along with the authorization Fact Sheet for Healthcare Providers, the authorized Fact Sheet for Patients and authorized labeling are available on the FDA website.
The Clarifi COVID-19 Test Kit has not been FDA cleared or approved. It has been authorized by the FDA under an emergency use authorization for use by authorized laboratories. Clarifi COVID-19 has been authorized only for the detection of nucleic acid from SARS-CoV-2, not for any other viruses or pathogens, and is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostic tests for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Act, 21 U.S.C. § 360bbb-3(b)(1), unless the authorization is terminated or revoked sooner.
This test kit is not a direct to consumer test. This test kit is sold only to clinical laboratories in quantity.
Quadrant Laboratories, a subsidiary of Quadrant Biosciences, is a CLIA certified laboratory that provides individual and pooled COVID testing services ordered by a licensed healthcare provider using the Clarifi COVID Test Kit sold by Quadrant Biosciences.
Note: Testing is limited to qualified laboratories in the United States, certified under the Clinical Laboratory Improvement Amendment of 1988 (CLIA), 42 U.S.C. SS 263A, to perform high complexity testing.